Background: CD74 is highly expressed on B cell malignancies, including non-Hodgkin's lymphoma (NHL). STRO-001, a novel CD74-targeting ADC was generated using cell-free protein synthesis and site-specific conjugation platform technologies. STRO-001 contains a potent maytansinoid warhead conjugated to two specific sites (drug-antibody ratio of 2) using a stable non-cleavable linker. This first-in-human Phase 1, open-label, multicenter, dose escalation study was designed to evaluate the safety, tolerability, and preliminary anti-tumor activity of STRO-001 in adults with B-cell malignancies (NHL and multiple myeloma). Herein we report preliminary results from the B-cell NHL cohort.
Methods: Patients with advanced, relapsed/refractory NHL are eligible for enrollment. STRO-001 is administered as a 60-minute IV infusion. STRO-001 was initially administered on Days 1 and 15 of a 28-day cycle. Starting at 0.91 mg/kg, STRO-001 was administered on Day 1 of a 3-week cycle. Treatment is administered until disease progression or unacceptable toxicity. The study employed a modified 3+3 design with an accelerated dose titration (N=1 per cohort until set specified AEs are observed) for initial dosing cohorts.
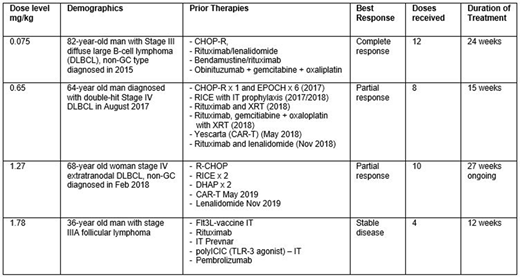
Results: 18 patients with NHL have been treated at 9 dose levels: .05, .075, .15, .27, .43, .65, .91, 1.27 and 1.78 mg/kg. NHL subtypes include: 6 diffuse large B-cell lymphoma (DLBCL), 5 follicular lymphoma (FL), 2 mantle cell lymphoma (MCL), 2 marginal zone lymphoma, 1 Burkitt's lymphoma, 1 composite DLBCL/FL and 1 composite DLBCL/CLL. Median age is 64.5 (range 21-82). Median ECOG performance status is 1 (range 0-2). Median number of prior therapies is 4 (range 1-12). Three patients received prior CAR-T therapy. Median number of STRO-001 doses administered is 2 (range 1-12). 17 patients have completed at least one cycle of STRO-001 and are evaluable for safety and toxicity for dose escalation recommendation. One patient at the 1.78 mg/kg dose level is currently completing Cycle 1 and not yet evaluable for DLT assessment. Most AEs are grade 1 or 2 (90%) with the most common grade 1-2 TEAEs of chills, fatigue, nausea, anemia, headache, pyrexia, infusion reaction, decreased appetite, and abdominal pain occurring in ≥ 20% of patients. There was one DLT in the NHL cohort, a grade 3 thromboembolic event at the 0.91 mg/kg dose level. 16 patients are evaluable for response. The preliminary clinical benefit/disease control rate for all patients is 25% (4/16) including 1 patient with complete response (CR) 2 with partial response (PR) and 1 with stable disease (Table). One patient with DLBCL treated at .075 mg/kg achieved a CR after 2 cycles (4 doses) and progressed after 12 doses (on study 24 weeks). A DLBCL patient treated at 0.65 mg/kg achieved a PR at Cycle 3 and progressed after 8 doses (on study 15 weeks). A DLBCL patient treated at 1.27 mg/kg who achieved a PR has received 10 cycles and remains on study after 27 weeks. Preliminary PK analysis of ADC shows exposure increased (Cmax from 0.39 to 19 µg/mL) and (AUC0-tlast from 0.6 to 71 h*µg/mL) as dose increased from 0.05 to 0.91 mg/kg.
Summary/Conclusion: STRO-001 is the first ADC generated with novel cell-free protein synthesis technology and site-specific conjugation to be tested in the clinic. STRO-001 has been well-tolerated. No ocular or neuropathy toxicity signals have been observed and the MTD has not been reached. Preliminary anti-tumor activity has been observed in this heavily pre-treated patient population, including two DLBCL patients who had previously progressed after a CAR-T (Table). The study continues to enroll patients in dose escalation. Next planned dose levels are 2.5 mg/kg and 3.5 mg/kg. This study is registered with clinicaltrials.gov identifier NCT03424603.
Shah:Verastim: Consultancy; Kite Pharma: Consultancy, Honoraria; Lily: Consultancy, Honoraria; Cell Vault: Research Funding; TG Therapeutics: Consultancy; Miltenyi Biotec: Honoraria, Research Funding; Celgene: Consultancy, Honoraria; Incyte: Consultancy. Popplewell:Pfizer: Research Funding; Novartis: Research Funding; Roche: Research Funding. Andreadis:Gilead/Kite: Consultancy; Merck: Research Funding; Incyte: Consultancy; Karyopharm: Honoraria; Jazz Pharmaceuticals: Honoraria; Genentech: Consultancy, Current equity holder in publicly-traded company; BMS/Celgene/Juno: Honoraria, Research Funding; Novartis: Research Funding. Melear:AstraZeneca: Speakers Bureau; Janssen: Speakers Bureau. Spira:Cardiff Oncology: Research Funding; Takeda: Consultancy; Novartis: Consultancy; Merck: Consultancy; BMS: Consultancy; Incyte: Consultancy; Janssen: Consultancy; ADCT: Research Funding. Manda:AbbVie: Other: Investigator in AbbVie-sponsored clinical trials. Burke:Roche: Consultancy; AbbVie: Consultancy; Bayer: Consultancy; Astra Zeneca: Consultancy; Verastem: Consultancy; Morphosys: Consultancy; Adaptive: Consultancy; Epizyme: Consultancy; Kura: Consultancy; Celgene: Consultancy; Adaptive Biotechnologies: Consultancy; Bristol Myers Squibb: Consultancy; Gilead: Consultancy; Seattle Genetics: Speakers Bureau. Sharman:TG Therapeutics: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Pharmacyclics: Consultancy, Research Funding; AstraZeneca: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Acerta: Consultancy, Research Funding; Roche: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Bristol Meyers Squibb: Consultancy, Research Funding; BeiGene: Research Funding. Krishnan:Sanofi: Consultancy; Sutro: Membership on an entity's Board of Directors or advisory committees; Amgen: Speakers Bureau; Takeda: Speakers Bureau; BMS/Celgene: Consultancy, Other: Stock BMS, Speakers Bureau; Janssen: Consultancy; Regeneron: Consultancy; Z Predicta: Membership on an entity's Board of Directors or advisory committees. Shah:BMS, Janssen, Bluebird Bio, Sutro Biopharma, Teneobio, Poseida, Nektar: Research Funding; GSK, Amgen, Indapta Therapeutics, Sanofi, BMS, CareDx, Kite, Karyopharm: Consultancy. Kuriakose:Sutro Biopharma: Current Employment. Berman:Sutro Biopharma: Current Employment. Matheny:Sutro Biopharma: Current Employment. Leonard:Miltenyi: Consultancy; BMS/Celgene: Consultancy; Regeneron: Consultancy; Karyopharm: Consultancy; GenMab: Consultancy; Sutro: Consultancy; Roche/Genentech: Consultancy; Epizyme: Consultancy; Bayer: Consultancy; Gilead/Kite: Consultancy; ADC Therapeutics: Consultancy; MEI Pharma: Consultancy; AstraZeneca: Consultancy. Molina:Sutro Biopharma: Current Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal